Antioxidants are substances that aid in the protection of the body from damaging molecules known as free radicals. Free radicals can be produced naturally in the body or obtained from external sources such as pollution, cigarette smoke, and some drugs. Antioxidants neutralize these free radicals, reducing oxidative stress and cell damage. Vitamins C and E, beta-carotene, selenium, and flavonoids are examples of common antioxidants. They are found in a wide range of foods, including fruits, vegetables, nuts, and seeds.
What antioxidants do in our body?
Antioxidants are substances that help protect our cells from free radicals, which are destructive compounds. Free radicals are highly reactive and can produce oxidative stress, which has been related to a variety of diseases and the ageing process. Antioxidant-rich foods and supplements can neutralize free radicals by donating an electron without becoming unstable themselves. This helps to prevent or minimize oxidative stress and potential cell and DNA damage. Antioxidants contribute significantly to overall health and well-being as follows

- Protecting against chronic diseases: By preventing cellular damage produced by free radicals, antioxidants can help reduce the risk of chronic disorders such as heart disease, cancer, and neurological diseases.
- Boosting the immune system: Antioxidants help to maintain a healthy immune system by shielding immune cells from oxidative stress, allowing them to operate efficiently in battling infections and illnesses.
- Supporting skin health: Antioxidants like Vitamins C and E, for example, help protect the skin from oxidative damage produced by UV radiation and environmental contaminants, promoting a young appearance and lowering the risk of skin illnesses.
- Enhancing eye health: Antioxidants like lutein and zeaxanthin build up in the eye and help protect it from oxidative damage, lowering the risk of age-related macular degeneration and cataracts. Lutein and zeaxanthin are antioxidants that protect your cells from harm. Most significantly, they aid in the removal of free radicals from your eyes.
- Supporting brain function: Cognitive decline and neurodegenerative illnesses can be exacerbated by oxidative stress. Antioxidants, especially those present in fruits and vegetables, have been demonstrated to have neuroprotective properties and may aid in brain health maintenance.
While antioxidants have numerous potential health benefits, they should be ingested as part of a well-balanced diet rather than as supplements. Eating a variety of fruits, vegetables, whole grains, nuts, and seeds can give a variety of antioxidants that work together to enhance our health.
Plant-based antioxidant compound
There are numerous antioxidant compounds found in plants. Some of the commonly known ones include Resveratrol, Quercetin, Anthocyanins, Isoflavones, Catechins, Lycopene, etc
How antioxidant compound neutralizes free radicle
The method of antioxidant activity varies depending on the molecule, but in general, antioxidants work by giving an electron or a hydrogen atom to the free radical, stabilizing it, and preventing further damage.
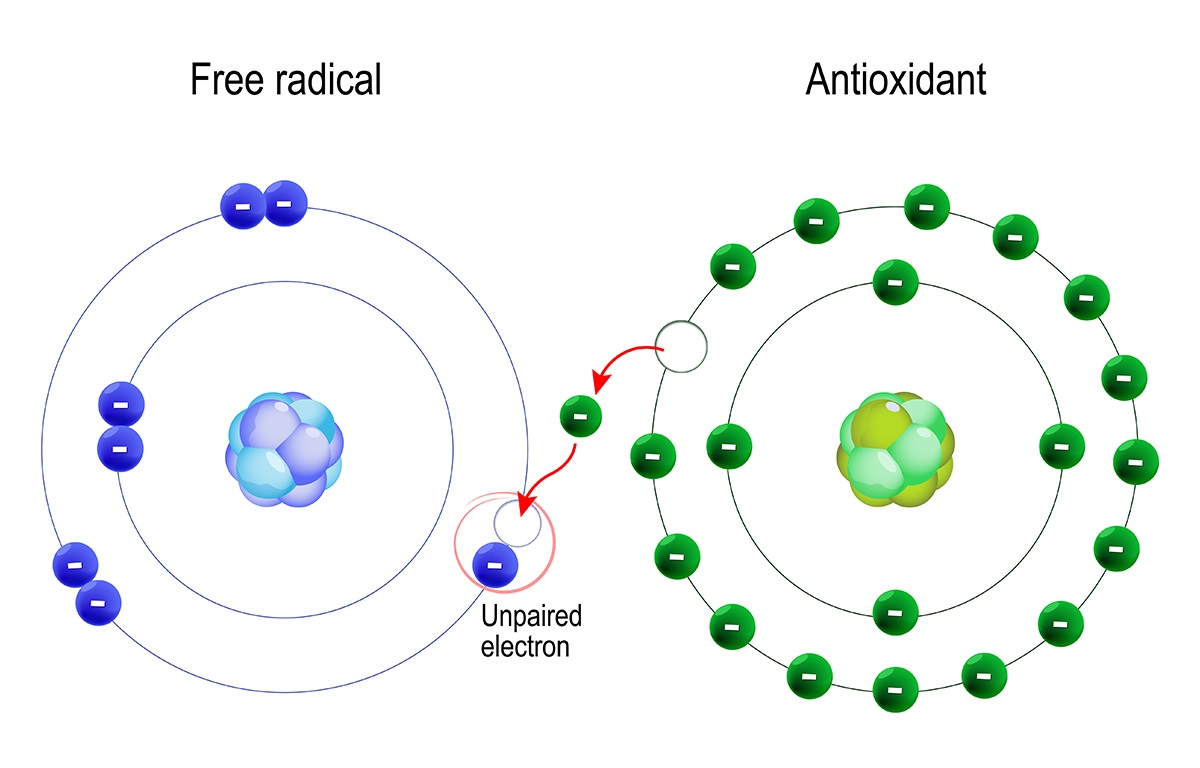
There are several ways in which antioxidants can carry out this mechanism:
- Direct scavenging: Some antioxidants react with free radicals directly, neutralising them by giving an electron or a hydrogen atom. This contributes to the breakdown of the chain reaction of free radical production.
- Indirect scavenging: Some antioxidants work indirectly by regenerating other antioxidants that have been oxidized by free radicals. For example, vitamin C can regenerate vitamin E, which is an important antioxidant in cell membranes.
- Metal chelation: Metal chelating agents are exhibited secondary antioxidants. However, some antioxidants can bind to metal ions, such as iron and copper, which can otherwise generate reactive oxygen species (ROS) through Fenton reactions. By chelating these metal ions, antioxidants prevent the formation of ROS.
- Enzyme regulation: Some antioxidants can regulate the activity of enzymes involved in the production of reactive oxygen species. For example, superoxide dismutase (SOD) is an enzyme that converts superoxide radicals into less harmful molecules, and antioxidants can enhance its activity.
Overall, antioxidants act by reducing the levels of reactive oxygen species and minimizing their harmful effects on cells and tissues. They play a crucial role in maintaining oxidative balance in the body and protecting against oxidative stress-related diseases.
Which methods are used for antioxidant activity tests?

The ability of a substance to neutralize or limit the detrimental effects of free radicals in the body is determined by an antioxidant activity test. There are several ways for evaluating antioxidant activity, including:
- Oxygen Radical Absorbance Capacity (ORAC) assay: This test evaluates a substance’s ability to scavenge and neutralise oxygen free radicals.
- 2,2-diphenyl-1-picrylhydrazyl (DPPH) assay: This assay employs a stable free radical, DPPH, to assess a substance’s capacity to donate hydrogen atoms and neutralize the free radical.
- Ferric Reducing Antioxidant Power (FRAP) assay: This test determines a substance’s reducing capability by evaluating its ability to convert ferric ions to ferrous ions.
- Trolox Equivalent Antioxidant Capacity (TEAC) assay: This assay compares a substance’s ability to scavenge free radicals to the antioxidant activity of Trolox, a synthetic version of vitamin E.
Thus, all of these tests are often conducted in a laboratory setting and can provide valuable information about the antioxidant potential of a substance or product.
What Cultivator Phyto lab can do for an antioxidant activity test
Cultivator Phyto Lab is an innovative laboratory that provides a wide range of chemical testing and biological services. We have developed multiple methods to measure antioxidant activity in a wide range of matrices and food commodities, including AYUSH and agricultural goods.
Authors: Dr. Sanjoy Gupta and Sajid Hussain
Reference:
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3705341/pdf/nutrients-05-01169.pdf
- BLOIS, M. Antioxidant Determinations by the Use of a Stable Free Radical. Nature 181, 1199–1200 (1958). https://doi.org/10.1038/1811199a0
- https://www.jocpr.com/abstract/antioxidant-activity-of-different-parts-of-lysimachia-laxa-and-gymnocladus-assamicus-a-comparison-using-three-different–1734.html
- https://link.springer.com/article/10.1007/s13205-017-0907-2
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6076941/
- https://www.sciencedirect.com/science/article/abs/pii/S0278691502001692?via%3Dihub
- https://www.frontiersin.org/articles/10.3389/fenvs.2014.00053/full
- https://link.springer.com/article/10.1007/s12013-009-9043-x

